
Computer System Validation Process (CSV)
Computer System Validation (CSV) is a systematic and documented process used in highly regulated industries such as pharmaceuticals to ensure that computerised systems (software and hardware) consistently perform their intended functions in a reliable, accurate and compliant manner. The primary goal of CSV is to demonstrate that these systems are fit for their intended use and adhere to regulatory requirements..
It also ensures data integrity, traceability and compliance with regulatory agencies such as the U.S. Food and Drug Administration (FDA-CFR21 Part 11) and the European Union’s Good Manufacturing Practice (GMP – Annexure 11).
In the pharmaceutical industry, any computers (software and hardware) involved in the following processes should be validated; R & D, laboratory lab notebook manual, batch record manufacturing process, analytical testing, stability process and material storage & packaging. Therefore, CSV is a stringent requirement as per national and international guidelines for the pharma industry at large..
Our Validation Services
We ensure that your computer systems are compliant
We have a high level of expertise in both compliance and project management. This enables us to ensure that computerised systems are compliant at all levels and that compliance processes are run efficiently. Furthermore, we are experienced in all parts of the validation model: From planning, risk-based validation, training and validation related documentation.
We provide Risk Assessments, Software Vendor Audits, Validation Plans, Reports and More
At Robosol, we have a high level of expertise in Data Migration, User Requirement Specifications, System Management SOPs and much more. This spectrum of expertise enables us to deliver the best and most in-depth consulting services.
Data disaster recovery, data sync, replication and failover groups are some of the important aspects of any validation task for which Robosol with our expert consultants is ready to assist you to ensure that your data is readily available and reliable.
System Validation Process
Computer system validation broadly requires the validation plan, training and documentation including standard operating procedure (SOP) as detailed in the process diagram shown below.
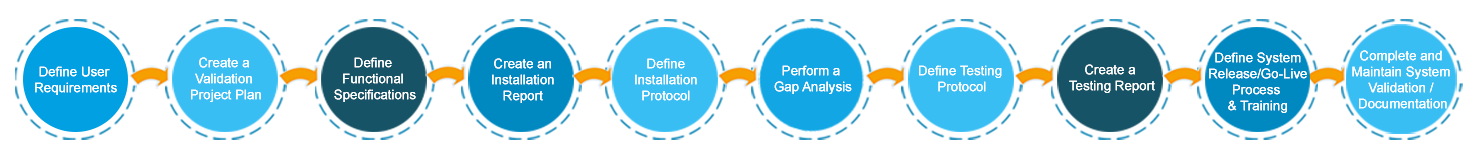
Validation Process Flow
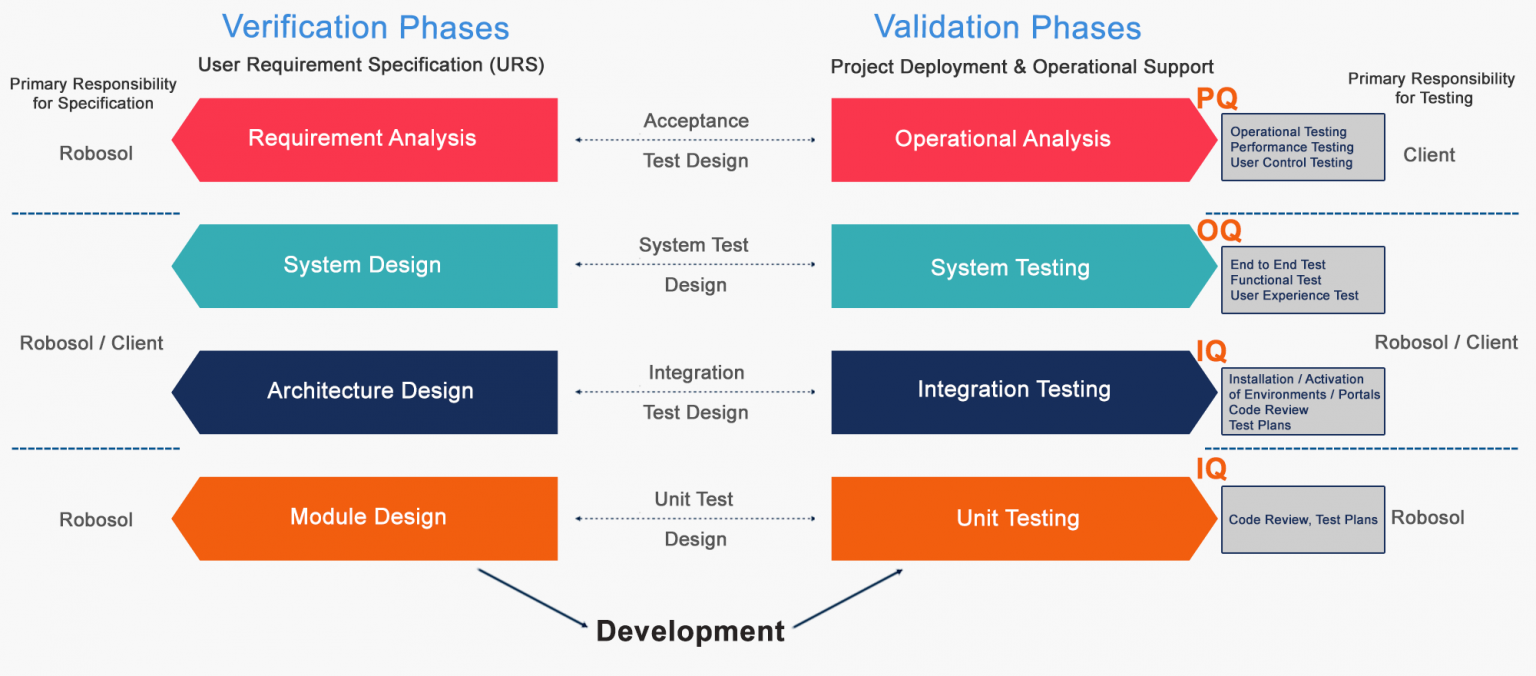
The diagram below describes the relationship between each phase of the development
life cycle and its corresponding testing phases
Download Computer System Validation Brochure
System Verification Stages
The IQ verifies that the requirements defined in the document are installed as originally specified in Company’s Functional Design Document.
- Tenant Qualification
- Environment Setups
- Disaster Recovery and Security
- Authentication and User Permissions
- Software Access on Desktop
- Mobile devices
This OQ protocol will provide the necessary documented verification that all key aspects of the Microsoft Dynamics 365 Business Central have been properly tested.
- Customising Dynamics BUSINESS CENTRAL
- Personalising the Workspace
- Set Filter
- Displaying lists
- Sort Records
- Search for page or report
- Export Data
- Event Log & Audit Trial Verification
- Security Preference Verification
Performance Qualification protocol will provide the necessary documented verification that all key aspects of the Microsoft Dynamics 365 Business Central have been properly tested.
- Test Script Execution and Dependency Summary
- Test Script Reference Material (usernames, passwords, product information etc.)
- Business Central Credentials
Typically during an ERP implementation there are various configurations and customisations made during the design specification. It is imperative to ensure that there is consistency in the approach and execution of the design, testing and deployment method outlined throughout the ERP Implementation. The Validation process is there to aid this process and provide the necessary governance to ensure the deployment is done in a controlled manner and conforms to the necessary regulatory guidelines as outlined by FDA (U.S. Food and Drug Administration) or GMP (European Union’s Good Manufacturing Practice) or any other relevant regulatory bodies to ensure compliance.
Robosol, as an certified ERP service provider, plays a pivotal role in bolstering our clients compliance endeavours by offering software solutions that adhere to regulations owing to our streamlined validation processes.
Click here to learn more about our offerings for Pharmaceutical Sector
To know more about how we can help you validate your systems Click here

